- Home
- About
- Contact
- How to do dilution series
- Free adobe photoshop cc 2017
- Best modded whatsapp 2018
- Element 3d v2-2 elemnt failed initialize opengl
- Alex p- nguyen oncology consultants pa
- Hustler magazine covers 1981
- Floor plan vr free donwload
- Igi 2
- Physicus save the world with science download
- Downloading kik for pc
- Sankat mochan mahabali hanumaan
- Flow the psychology of optimal experience audiobook free
- Nes emulator with controller support mac
- Adventures of merlin season 6
- Shabad kirtan lyrics in punjabi
- Holdem manager 2 americas cardroom not importing hands
- First touch soccer 2015 aptoide
- Music from happy days movie download
- Synchro resolver tutorial
- Download rock band unplugged dlc
- Microsoft flight simulator x gold edition download
- Ufps vs realistic fps prefab
- #HOW TO DO DILUTION SERIES SERIAL#
- #HOW TO DO DILUTION SERIES MANUAL#
- #HOW TO DO DILUTION SERIES SOFTWARE#
- #HOW TO DO DILUTION SERIES SERIES#
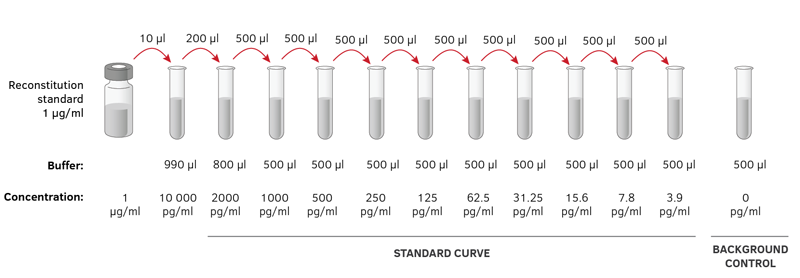
Repeat the process until you have four tubes. Then transfer 0.2 mL from Tube 2 to 3.8 mL of diluent in Tube 3 and mix. You would transfer 0.2 mL from Tube 1 to 3.8 mL of diluent in Tube 2 and mix. So you multiply each successive dilution by the dilution factor.
#HOW TO DO DILUTION SERIES SERIAL#
Remember that serial dilutions are always made by taking a set quantity of the initial dilution and adding it successively to tubes with the same volume. If you did the above dilution four times, what would be the final dilution factor? What is the dilution factor if you add 0.2 mL of a stock solution to 3.8 mL of diluent? The dilution factor or the dilution is the initial volume divided by the final volume.įor example, if you add a 1 mL sample to 9 mL of diluent to get 10 mL of solution, In serial dilutions, you multiply the dilution factors for each step. With this non-contact direct dilution, less compound material is required, saving precious compound material and cutting consumable costs.You multiply the original concentration by the dilution factors for each dilution.Ī serial dilution is any dilution in which the concentration decreases by the same factor in each successive step.

#HOW TO DO DILUTION SERIES SOFTWARE#
Normalization with buffer or solvent can also be easily set up with the implemented software to adjust all wells to the same volume. In contrast to serial dilution, direct dilution transfers different volumes of the same stock solution directly into successive wells to generate the required dilution series. Importantly, the method does not rely on traditional plastic pipette tips, thus enabling direct dilution.

The technology allows for fast dispensing of low volumes, ranging from single digit nanoliters up to several microliters. Using the I.DOT One non-contact dispenser, these limitations can be avoided.
#HOW TO DO DILUTION SERIES SERIES#
Moreover, these methods often not allow for preparing microvolume dilution series in pure DMSO, which can be problematic for small molecule compounds. The most striking drawback of this traditional way is error propagation, which increases with each transferring step in the serial dilution due to deviations in pipetting accuracy and precision. This procedure is continued and finally results in a gradually lower reagent concentration. This is done by transferring small amounts of high concentrated stock reagents (in DMSO or buffer) from one well to the next. Traditionally, multichannel pipettes and pipetting robots prepare the required dilution series across a number of wells in microplates.
#HOW TO DO DILUTION SERIES MANUAL#
The binding affinity, determined by a curve-fit, agrees well between the experiment with the manual generated compound serial dilution and the two experiments with the I.DOT One generated compound serial dilution. Manually generated data (red) are compared to the I.DOT One performance (light green: first run, dark green: second run).

In such an assay the binding affinity of a compound to a protein target is analyzed by testing a serial dilution of the compound. The example curves below show the readout of a MicroScale Thermophoresis (MST) binding assay performed by 2bind GmbH. Here, serial dilution series are required to record dose-response curves and to calculate the steady-state affinity, e.g. Particularly, the determination of steady-state binding affinities is widely used in biochemistry and pharmacology for drug discovery processes. Dilution series are a common method in different scientific fields to generate standard curves for calibration of instruments or for the evaluation of the bioactive potential of diverse substances.
